
FAQ
The most frequently asked questions
FAQ
Frequently asked questions about clinical trials:
Below you will find an overview of the most frequently asked questions about Swiss-GO's clinical cancer research studies.
We hope that you will find the right answer to your query directly. If not, please contact your doctor or email
A clinical trial is a research project that examines how well a new treatment or medical procedure works in humans. Other studies that are performed only on cells in petri dishes or on animals are referred to as preclinical studies. Clinical studies only begin after preclinical studies have shown that the new treatment or procedure helps people and is also safe for people.
Preclinical studies offer researchers a lot of good information. However, studying how a treatment or procedure works in humans is different from studying mice or specific cells in a laboratory. Therefore, clinical studies are required to answer important questions:
- Does the new treatment or procedure work in humans?
- Is what is being tested better than what is being used now?
- Does it cause more or less side effects?
- Does it work in a group of people who can or cannot use current treatments or procedures?
- Is the new treatment or procedure safe for people?
All treatments for cancer and some diagnostic procedures have side effects. In clinical trials, researchers are trying to find out whether the benefits of a new treatment or a new procedure outweigh the possible side effects.
Researchers carry out clinical studies in different environments. Many clinical trials are carried out in cancer centers at academic hospitals, because the facilities available there are usually the most suitable ones. Nowadays, general hospitals and medical practices can also be part of a clinical trial.
This has broadened the possibility for patients to take part in a trial. Instead of going to a non-familiar hospital and getting treatment from an unknown doctor, patients can participate in clinical trials and still be treated by their own doctor (as long as he or she is involved in the study). The research team that carries out a clinical trial consist mostly of scientists, doctors, nurses, social workers, dietitians, and other health professionals.
Clinical trials can be funded by pharmaceutical companies or non-profit organizations.
In Switzerland, the Swiss Human Research Act (HFG) regulates human research. A study can only be carried out if it has been checked and approved by an independent control body. In Switzerland, two control bodies are in place, the Ethics Committees and the Health Authority Swissmedic.
The researchers who plan to start a clinical trial write a plan as to how the trial should be carried out to find the right answers to the questions; is this drug or procedure safe, effective, and/or superior to the current treatment options. This plan is called the protocol. The protocol is a very technical document and contains details of the dosage of each treatment and the frequency and duration of each treatment. It also lists all laboratory work and tests that need to be done to be able to find out how each person in the study responds to treatment.
The protocol is reviewed by both control authorities that either approve or reject the protocol and subsequent study. The control authorities take several questions into account when making their decision:
- Is it ethical to ask people to volunteer for this experimental treatment?
- Was the study developed to ensure that the people involved are safe?
- Will participants receive treatment that is at least as good or better than what they would receive if they did not participate in the study?
This supervisory body is composed of scientists, doctors, nurses, ethics, law and patient representatives who are not involved in the clinical study. If the clinical trial is approved, the review board will monitor the study and formally review it each year.
After the protocol has been approved, the researchers ask participants to participate in the study.
If you want to participate in a clinical trial, the first step is to contact your doctor. He or she may know a clinical trial that is open and suits your particular situation best.
If you want to make sure a study is relevant for you, you additionally can call the nearest cancer center and ask for information about this study. You can also search for clinical trials on the Centre's website or on the Swiss-GO, SAKK or ENGOT websites in relation to gynecological cancer.
On the portal of the Federal Office of Public Health for Human Research in Switzerland you can find additional information on approved studies in Switzerland. You can also view clinical trials conducted worldwide from the American National Institute of Health's international registry https://clinicaltrials.gov or specific for Europe https://www.clinicaltrialsregister.eu
Because, researchers want to make sure that the results obtained during a clinical trial can be attributed to the new treatment or process and are not caused by coincidence, the participants of a clinical trial are being selected on very specific characteristics, the so-called eligibility criteria. For example, a study in which the effectiveness of an hormone inhibitor will be assessed in ovarian cancer will select their participants with the following characteristics:
- postmenopausal women
- diagnosed with hormone receptor positive ovarian cancer
- have had surgery to remove the cancer
- may or may not have received chemotherapy
One person must meet ALL admission requirements to participate in the study, the so-called inclusion criteria.
Part of the process is to enroll patients who are similar in certain points. For example, a study could serve to answer questions about the treatment of patients who have a certain stage of cancer or who have already received a certain type of chemotherapy. In order for the results to make sense at the end of the study, only those patients who meet these criteria are included.
Another aspect is patient safety. For example, a new drug can only be safe in people with normal kidney or liver function. Therefore, people with poorly functioning kidneys or livers would not be included in the study, they are so-called exclusion criteria.
So patients can only be included when they meet all inclusion criteria and none of the exclusion criteria. This will be assessed by the physician or study nurse prior to enrolment in a clinical trial.
A participant must give a so-called declaration of consent (also known as informed consent) before participating in a clinical trial. Upon consent, researchers must explain the clinical trial protocol to potential participants. Researchers must also inform potential participants about the potential risks and benefits of participating in the study. A person can then decide whether or not to participate in the study. It is also important to know that people can end a clinical trial at any time (long time) before a clinical trial starts.
Each clinical trial has its own benefits and risks depending on the type and aim of the trial. However, some potential benefits and risks are common to almost all clinical trials.
Possible benefits
- Access to treatment that is not yet available. This treatment can be more effective or have fewer side effects than the treatments currently available.
- Regular and thorough care from some of the best cancer doctors. The research team that conducts clinical trials typically consists of top physicians and scientists. Due to this close monitoring, possible side effects are noticed early and remedied immediately.
- Contribution to research that can save lives in the future. The cancer treatments we have today are available because a large number of women have chosen to participate in clinical trials to test them in the past
- The positive feeling when you play an active role in your own care. The decision to participate in a clinical trial can sometimes make patients feel more in control of their situation, which can lead to an increase in positive outlook and subsequently a better quality of life
Possible risks
- You cannot decide which treatment to receive. In randomized trials, you are randomly selected to receive a particular treatment. Some studies may give you a placebo (sugar pill). In a randomized, double-blind study, neither you nor your doctor knows if you are selected to receive the treatment or the placebo (but the information can be made available to your doctor if necessary).
- The new treatment may not work for you, even if it benefits other people in the study. It may also turn out that the new treatment is not as effective as the one currently available.
- More serious side effects than current treatments. This is more likely in phase I or phase II studies (See section on study phases in FAQ).
- Your insurance may not cover all costs. Be sure to contact your insurance company and one of the clinical trial coordinators so that you know exactly if additional costs are to be expected BEFORE agreeing to participate in a clinical trial.
- An increase in testing and doctor visits. Because you are closely monitored, you may need to take more tests or more often than when not participating in the study. This could result in more time and travel to the doctor's office or hospital.
What do I definitely need to know before I decide to participate in a clinical trial?
The decision to participate in a clinical trial is very personal. The answers to the following questions by your doctor might help to get a better understanding of the options:
- Why do you propose this study?
- Why do you think this new treatment will be effective?
- Which kind of study phase is it?
- Has this treatment been tested before?
- What were the results of previous studies?
- Can I speak to someone who is already in the study?
- Who pays the trial costs?
- What are the possible treatments I can get? How often are they given?
- What types of tests do I need to have and how often will they be assessed?
- How will participating in this study affect my daily routine?
- What are the expected side effects?
- Is my treatment free of charge? Will my insurance cover the costs? What exactly do I have to pay?
- How long does the screening process take? and what is the total duration of the clinical trial?
- Is long-term follow-up part of the study? What does it include?
- If the treatment works for me, can I continue to receive it after completing the study?
- How do I get access to the results of the study?
After the clinical trial protocol has been approved by the national health care and ethic authorities, the researchers ask participants to participate in the study. Clinical trials typically have between one and four different treatment schemes, the so-called treatment arms. In most cases, people who enroll in a clinical trial are randomly assigned to one of the treatment arms. This is known as a "randomized" study.Many clinical studies are "double blind". This means that neither the participant nor the researcher know which treatment arm the participant has been assigned to. Double-blinded clinical trials help researchers understand the real benefits and side effects of treatment without distortion of external influences. In this context, external influences mean any preconceived notions about treatment that may affect the way you take the medicine, which in turn may affect the results of the study.
The medication you receive is labeled with a code that is kept confidential by a small group of people who are not involved in your treatment. The code will be released at the end of the study when the results are evaluated. Whenever possible, clinical trials are both randomized and double-blinded as results obtained this way are regarded the most reliable.
Clinical trials are conducted to collect data regarding the safety and efficacy of new medication, health care processes and/or devices. There are several steps and stages of approval in the clinical trial process before a medication, process or device can be implemented in the regular treatment guidelines of a particular disease.
Medication, process or device development usually starts with the search for an improved treatment option with better outcome than the one in place. This development begins with extensive laboratory research, which can take many years of experiments in animal and human cells. If the initial laboratory research is successful, researches send the data to the competent healthcare authorities to obtain approval to continue the research in humans.
After approval, human testing can start and is mostly conducted in four phases (for medication, and at least three for devices and processes). Each phase is considered a separate trial and, after completion of a phase, investigators are required to submit their data again for approval to the competent authorities, before moving to the next phase. In this way, each step or phase builds on the results of the previous phase (see figure for visualization).
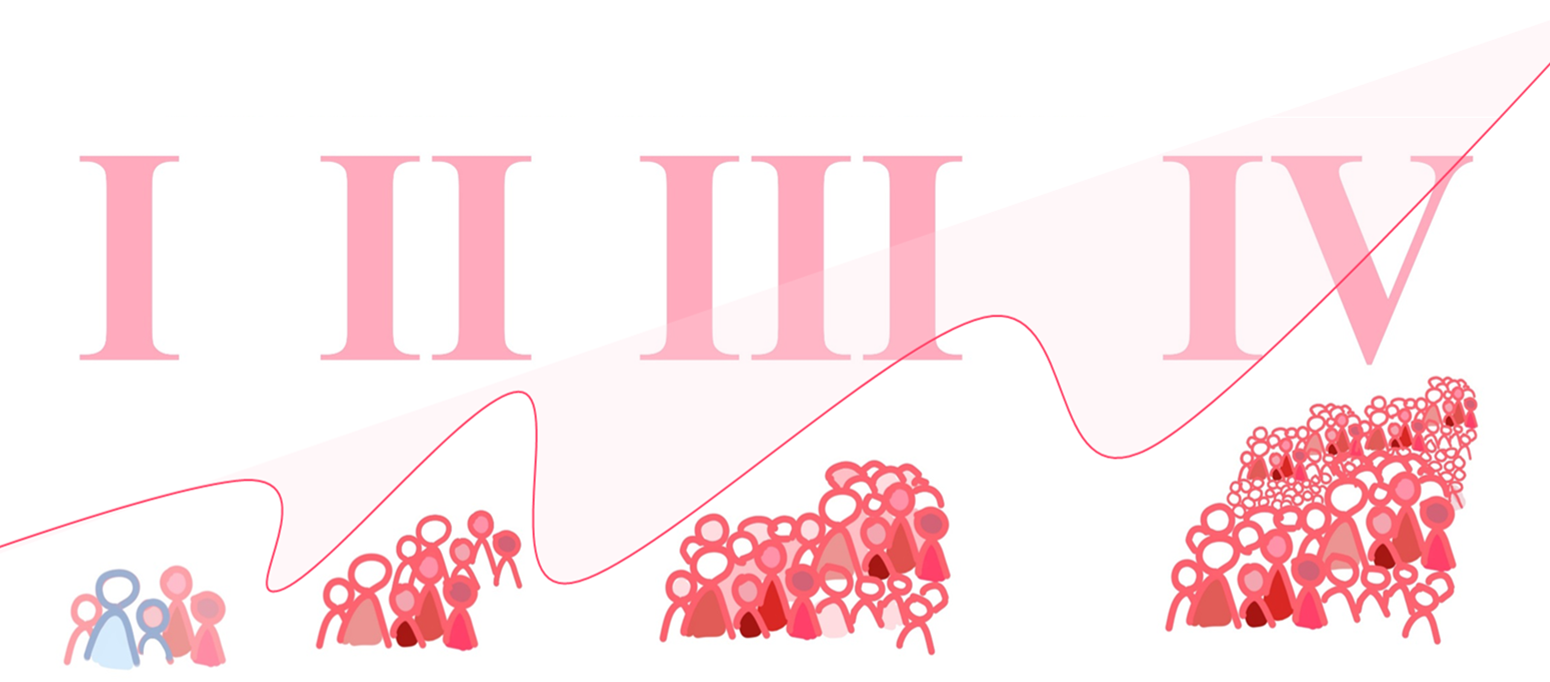
Phase I: is it safe?
Phase I trials assess the safety of a drug process or device. It is the first phase that involves humans. (Preclinical studies on petri dishes and animals have already been carried out.) Phase I studies are small and usually involve between 15 and 50 healthy volunteers. In phase I studies, researchers investigate:
- the best way to give a new treatment (as an injection or as a pill)
- how the drug is absorbed, metabolized and excreted in humans
- the highest dose that can be administered safely without serious side effects.
In phase I studies, researchers closely monitor participants and titrate the dose so they can define the amount that works best with the least acceptable side effects. This dose is usually used for all future tests in the subsequent phases.
Phase II: does it work?
Phase II studies test the efficacy of the new intervention. Phase II trials are slightly larger than Phase I trials and typically involve 25 to 200 patients. Most phase II studies are randomized trials where one group of patients receives the experimental drug, while a second "control" group receives a standard treatment or placebo. Researchers start with the dose and method proved best in Phase I. Phase II patients receive the new treatment and researchers observe whether the treatment is beneficial. The benefits that researchers are looking for may vary and depends on the objectives of the research:
- tumor is getting smaller
- tumor stops growing
- an increase in time before the cancer comes back
- longer survival time
- better quality of life
If a certain percentage of participants benefit from the treatment and the side effects are still acceptable, the new treatment will likely move to phase III.
Phase III: is it better than the current treatment options?
Phase III studies compare the safety and effectiveness of the new treatment with the current standard of care. Phase III studies are typically large (some include tens of thousands of participants) and are conducted at many locations (multi-centered) and often in different countries around the world (European, International, Global). A phase III trial is the final step before the competent authorities consider approval and grant market access.
Patients are usually randomly selected to receive the current standard treatment or the new treatment. If possible, the study is double-blinded, meaning that neither the researchers nor the participants know who receives which treatment. Double-blinded clinical trials help researchers understand the real benefits and side effects of treatment without distortion by external influences. Results from a randomized, double-blinded study are considered more credible than results from a non-randomized or double-blinded study.
As in Phase I and Phase II studies, participants in a Phase III trial are closely monitored to determine if there are any serious side effects. Treatment is stopped if side effects appear dangerous.
Phase IV: is it still beneficial over a longer period of time?
Phase IV studies, also called Post Marketing Surveillance Trials, typically review whether the treatment offers benefits or long-term side effects that have not been studied or observed during Phase II or Phase III. Phase IV studies are typically conducted after an intervention or treatment has been approved for use by the competent authorities. Phase IV studies are rarer than Phase I, II, or III studies and affect usually hundreds of thousands of patients addressing mainly the following:
- comparison to drugs already on the market or new treatment options.
- monitoring long-term effectiveness and impact on a patient's quality of life
- determine the cost-effectiveness relative to other traditional and new therapies.
Phase IV studies can result in a drug or device being taken off the market or restrictions of use could be placed on the product depending on the findings in the study.
All data collected during the clinical trial is subject to professional confidentiality. Data about you, your state of health and the treatment are recorded and stored so that they are available for evaluation. However, the data will only be passed on for this purpose in encrypted (anonymized) form. As a patient, your personal identity and traceability is protected during as well as after closure of the clinical trial.
Participation in a clinical trial is equivalent to treatment.
The clarification and treatment within a clinical trial does not incur any additional or special costs for you, even if the therapy is more expensive than the standard treatment. However, it might require additional time and travel expenses.
Patients considering participating in a clinical trial should talk with their doctor or medical caregivers. Potential participants should understand the credentials and experience of the staff and the facility involved in conducting the study.
The decision to participate, however, remains very personal, but it can be helpful to ask your doctor these general questions:
- How long will the trial last?
- Where is the trial being conducted?
- What treatments will be used and why do you think this new treatment will be effective?
- What is the main purpose of the trial?
- What phase is this study in?
- How does participating in this study affect my daily routine?
- How will patient safety be monitored?
- Are there any risks involved?
- What are the possible benefits?
- What are the alternative treatments besides the one being tested in the trial?
- Who is sponsoring the trial?<
- Do I have to pay for any part of the trial?
- What happens if I am harmed by the trial?
- Can I opt to remain on this treatment, even after termination of the trial?